 The current near-ubiquitous use of acetaminophen during pregnancy is due in part to the widespread perception – even among doctors – that it has limited side effects and negligible risk. More research is needed to determine whether this practice is safe for the developing brain. Notably, acetaminophen is also the medication most commonly given to infants. Additionally, we would like to see research that gives us a better understanding of the biologic pathways. But to really get at these questions, we need human cohort studies that can precisely capture when and why acetaminophen is taken during pregnancy. It’s unethical to do experiments that could harm a human life, so to gain a better understanding of the direct effects of acetaminophen during pregnancy, we must rely on human observational and experimental studies to assess the possibility of causal connections. We conclude that because acetaminophen is so commonly taken during pregnancy, if its use is responsible for even a small increase in individual risk, it could contribute substantially to these disorders in the overall population.  
Over the same time period, the use of acetaminophen during pregnancy has gone up. Rates of reproductive disorders and neurodevelopmental disorders, such as ADHD and autism spectrum disorder, have been increasing over the last 40 years. Research suggests that acetaminophen is an endocrine disruptor and may interfere with the hormones essential for healthy neurological and reproductive development.Ī spokeswoman for Johnson & Johnson, which makes Tylenol, told CNN in September that the product label tells women who are pregnant or breastfeeding to ask a health care professional before using it.Ĭurrent guidance recommends acetaminophen as the pain reliever of choice during pregnancy, as other pain relievers such as ibuprofen and aspirin are not considered safe after midpregnancy. It is used by more than 50% of pregnant women worldwide and at least 65% of pregnant women in the U.S. Why it mattersĪcetaminophen is an active ingredient in over 600 prescription and over-the-counter medications, including Tylenol. 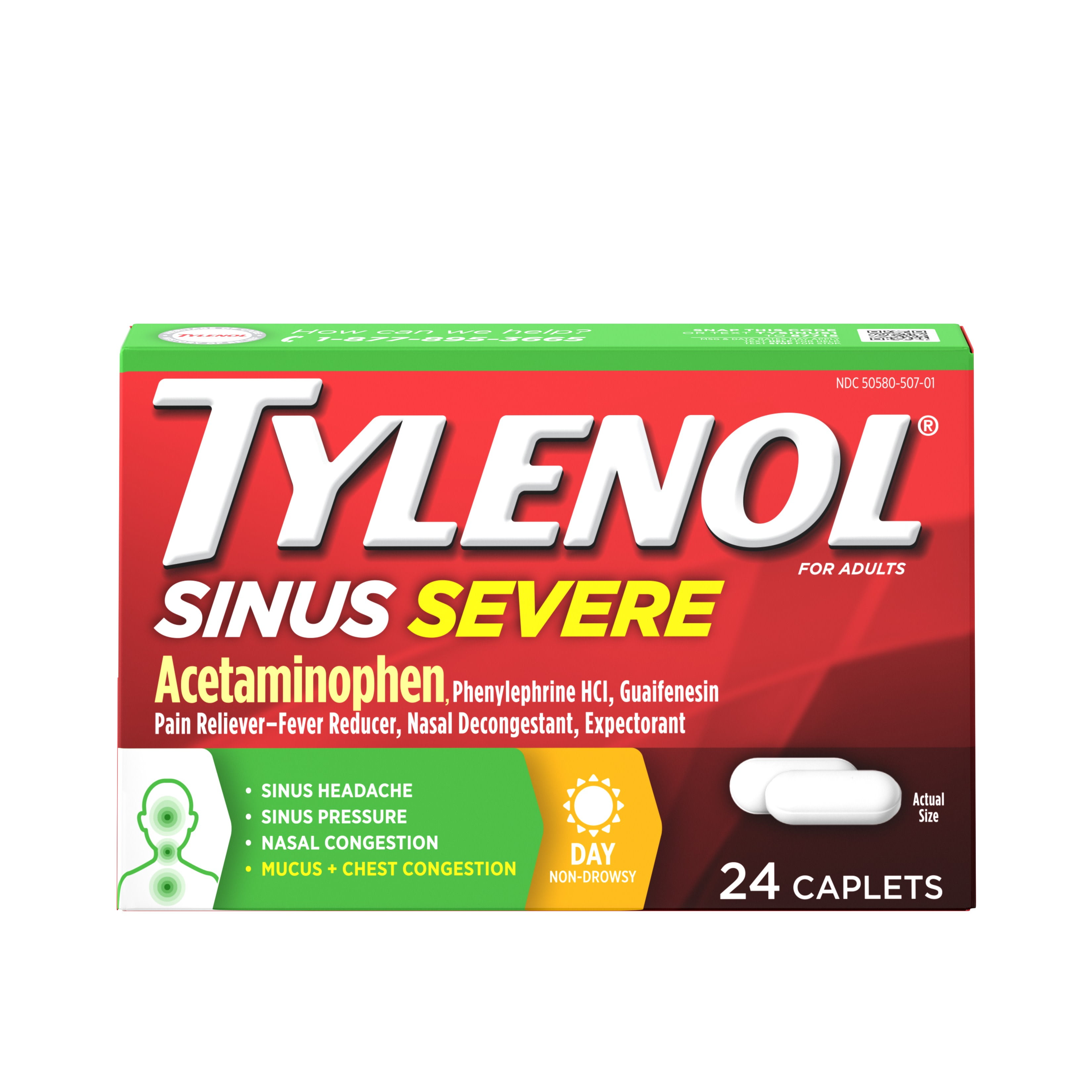
In our consensus statement – a broad agreement by our multidisciplinary international panel of experts – published in Nature Reviews Endocrinology in September 2021, 91 clinicians and researchers are calling for caution and additional research. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
